Ed Friedlander, M.D., Pathologist
scalpel_blade@yahoo.com
No texting or chat messages, please. Ordinary e-mails are welcome.

|

|
 |
 |
 |
 |
|
verify here. |
Cyberfriends: The help you're looking for is probably here.
This website collects no information. If you e-mail me, neither your e-mail address nor any other information will ever be passed on to any third party, unless required by law.
This page was last modified January 1, 2016.
I have no sponsors and do not host paid advertisements. All external links are provided freely to sites that I believe my visitors will find helpful.
Welcome to Ed's Pathology Notes, placed here originally for the convenience of medical students at my school. You need to check the accuracy of any information, from any source, against other credible sources. I cannot diagnose or treat over the web, I cannot comment on the health care you have already received, and these notes cannot substitute for your own doctor's care. I am good at helping people find resources and answers. If you need me, send me an E-mail at scalpel_blade@yahoo.com Your confidentiality is completely respected. No texting or chat messages, please. Ordinary e-mails are welcome.
 I am active in HealthTap,
which provides free medical guidance from your cell phone.
There is also a fee site at www.afraidtoask.com.
I am active in HealthTap,
which provides free medical guidance from your cell phone.
There is also a fee site at www.afraidtoask.com.
 If you have a Second Life account, please visit my teammates and me at the Medical Examiner's office. |

|
 | With one of four large boxes of "Pathguy" replies. |
 I'm still doing my best to answer
everybody.
Sometimes I get backlogged,
sometimes my E-mail crashes, and sometimes my
literature search software crashes. If you've not heard
from me in a week, post me again. I send my most
challenging questions to the medical student pathology
interest group, minus the name, but with your E-mail
where you can receive a reply.
I'm still doing my best to answer
everybody.
Sometimes I get backlogged,
sometimes my E-mail crashes, and sometimes my
literature search software crashes. If you've not heard
from me in a week, post me again. I send my most
challenging questions to the medical student pathology
interest group, minus the name, but with your E-mail
where you can receive a reply.
Numbers in {curly braces} are from the magnificent Slice of Life videodisk. No medical student should be without access to this wonderful resource.
 I am presently adding clickable links to
images in these notes. Let me know about good online
sources in addition to these:
I am presently adding clickable links to
images in these notes. Let me know about good online
sources in addition to these:
pathology.org -- my cyberfriends, great for current news and browsing for the general public
EnjoyPath -- a great resource for everyone, from beginning medical students to pathologists with years of experience
Medmark Pathology -- massive listing of pathology sites
Estimating the Time of Death -- computer program right on a webpage
Pathology Field Guide -- recognizing anatomic lesions, no pictures
Freely have you received, freely give. -- Matthew 10:8. My site receives an enormous amount of traffic, and I'm still handling dozens of requests for information weekly, all as a public service.
Pathology's modern founder, Rudolf Virchow M.D., left a legacy of realism and social conscience for the discipline. I am a mainstream Christian, a man of science, and a proponent of common sense and common kindness. I am an outspoken enemy of all the make-believe and bunk that interfere with peoples' health, reasonable freedom, and happiness. I talk and write straight, and without apology.
Throughout these notes, I am speaking only for myself, and not for any employer, organization, or associate.
Special thanks to my friend and colleague, Charles Wheeler M.D., pathologist and former Kansas City mayor. Thanks also to the real Patch Adams M.D., who wrote me encouragement when we were both beginning our unusual medical careers.
If you're a private individual who's enjoyed this site, and want to say, "Thank you, Ed!", then what I'd like best is a contribution to the Episcopalian home for abandoned, neglected, and abused kids in Nevada:

My home page
More of my notes
My medical students
Especially if you're looking for information on a disease with a name that you know, here are a couple of great places for you to go right now and use Medline, which will allow you to find every relevant current scientific publication. You owe it to yourself to learn to use this invaluable internet resource. Not only will you find some information immediately, but you'll have references to journal articles that you can obtain by interlibrary loan, plus the names of the world's foremost experts and their institutions.
Alternative (complementary) medicine has made real progress since my generally-unfavorable 1983 review. If you are interested in complementary medicine, then I would urge you to visit my new Alternative Medicine page. If you are looking for something on complementary medicine, please go first to the American Association of Naturopathic Physicians. And for your enjoyment... here are some of my old pathology exams for medical school undergraduates.
I cannot examine every claim that my correspondents
share with me. Sometimes the independent thinkers
prove to be correct, and paradigms shift as a result.
You also know that extraordinary claims require
extraordinary evidence. When a discovery proves to
square with the observable world, scientists make
reputations by confirming it, and corporations
are soon making profits from it. When a
decades-old claim by a "persecuted genius"
finds no acceptance from mainstream science,
it probably failed some basic experimental tests designed
to eliminate self-deception. If you ask me about
something like this, I will simply invite you to
do some tests yourself, perhaps as a high-school
science project. Who knows? Perhaps
it'll be you who makes the next great discovery!
Our world is full of people who have found peace, fulfillment, and friendship by suspending their own reasoning and
simply accepting a single authority that seems wise and good. I've learned that they leave the movements when, and only when, they discover they have been maliciously deceived.
In the meantime, nothing that I can say or do will
convince such people that I am a decent human being. I no longer answer my crank mail.
This site is my hobby, and I do not accept donations, though I appreciate those who have offered to help.
During the eighteen years my site has been online, it's proved to be one of the most popular of all internet sites for undergraduate physician and allied-health education. It is so well-known that I'm not worried about borrowers. I never refuse requests from colleagues for permission to adapt or duplicate it for their own courses... and many do. So, fellow-teachers, help yourselves. Don't sell it for a profit, don't use it for a bad purpose, and at some time in your course, mention me as author and William Carey as my institution. Drop me a note about your successes. And special thanks to everyone who's helped and encouraged me, and especially the people at William Carey for making it still possible, and my teaching assistants over the years.
Whatever you're looking for on the web, I hope you find it, here or elsewhere. Health and friendship!
BIBLIOGRAPHY / FURTHER READING
I urge anyone interested in learning more about diseases of blood clotting to consult these standard textbooks.
In my notes, the most helpful current journal references are embedded in the text. Students using these during lecture strongly prefer this. And because the site is constantly being updated, numbered endnotes would be unmanageable. What's available online, and for whom, is always changing. Most public libraries will be happy to help you get an article that you need. Good luck on your own searches, and again, if there is any way in which I can help you, please contact me at scalpel_blade@yahoo.com. No texting or chat messages, please. Ordinary e-mails are welcome. Health and friendship!
{26443} platelets
{26169} normal megakaryocyte
{13745} megakaryocyte
{25191} hematocele (guy got kicked probably)
{39557} hemorrhage into renal pelvis (this was a TTP case)
{05938} purpura from thrombocytopenia
|
|
|
 American Society of Hematology (?!)
American Society of Hematology (?!)
Image Bank
For the gung-ho students
![]() KCUMB Students
KCUMB Students
"Big Robbins" -- RBC / Bleeding
Lectures follow Textbook
QUIZBANK
Hemodynamic #'s 114-197
INTRODUCTION TO THE BLEEDING DISORDERS ("HEMORRHAGIC DIATHESES")
Your lecturer has no interest in presenting one more overview of the clotting cascades, the activation of platelets, or the mysteries of endothelium. We'll look today at diseases that affect one or more of these. I promise to keep it simple.
One more time... THE REAL CLOTTING CASCADE (i.e., how it works in life most of the time)
All diseases of inadequate hemostasis have spontaneous bleeding (petechiae, purpura, mucous membranes, GI bleeding, hematuria, into joint spaces, or even just unusually heavy periods -- the endometrium can be ablated for this (Am. J. Ob. Gyn. 202: 348.e1-7, 2010) and/or excessive bleeding after trauma or surgery.
The range is from lethal diseases (factor VIII:C deficiency, Bernard-Soulier's, Glanzmann's) to non-diseases (factor XII deficiency, many von Willebrand's).
Three groups:
TESTING HEMOSTASIS
Platelet count: reference range is 150,000 to 400,000 (or 450,000 or 350,000 or 300,000);
Platelets rise at ovulation, and fall low at the start of menstruation.
{14727} finger-stick blood; platelets clumped!
Bleeding time: checks both quantity and functional quality of platelets. Hard to standardize and not done that often.
Ivy: Stab with a 3 mm lancet. Normally bleeds 3-6 minutes.
Template: Use a standard spring-loaded razor device that produces a 1 mm deep, 3 mm long incision. Normally bleeds 6-10 minutes; probably this is longer than the Ivy time since platelet recruitment is slower for this tiny wound.
{14117} bleeding time, step 1
{14120} bleeding time, step 2
{14123} bleeding time, step 3
{14126} bleeding time, step 4
{14129} bleeding time, step 5
{14132} bleeding time, step 6
{14135} bleeding time, step 7
{14138} bleeding time, step 8
{14141} bleeding time, step 9
{14144} bleeding time, step 10
Thrombin time (TT): checks factor I (fibrinogen) activity. It's also the way we assess therapeutic range for dabigatran (the new oral thrombin inhibitor).
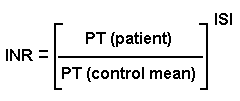
Prothrombin time (PT): checks activities of factors I, II, V, VII, and X
* You'll try to maintain your patients on coumarin in various ranges (2.0-3.0 to prevent deep vein thrombi in patients at high risk; 2.5-3.5 for patients with mechanical heart valves; etc., etc. Chest 108(S): 231, 1995).
Point-of-care testing to get the INR using fingerstick is now widely available; it delivers faulty values in patients with heparin or lupus anticoagulant on board.
It takes four days after starting warfarin for INR testing to be meaningful.
Heads up: If the phlebotomy is difficult, the little bit of tissue factor that gets into the blood before the anticoagulant can do its work is enough to deplete the clotting factors and cause a greatly prolonged INR. This explains the occasional person who needs to be called back for a repeat test. See Am. J. Clin. Path. 134: 86, 2010. Of course, if the tube has partially clotted (poor mixing), or the patient is heparinized from dialysis, or the specimen is drawn downstream from an intravenous fluid site, or the phlebotomist did not fill the tube completely or mix it properly, there's also likely to be a falsely long INR.
Activated partial thromboplastin time (aPTT): checks activities of factors I, II, V, VIII, IX, X, XI, XII. It is prolonged when one of these drops below about 30% of healthy. It's also prolonged by antibodies against phospholipid.
* A few novel tests (including one based on snake venom) are available for patients receiving the new short-acting direct thrombin inhibitors -- the big idea is that these are used during procedures and need not be monitored, but since they're lost through the kidneys and some folks have kidney failure, etc., testing may be necessary. See Am. J. Clin. Path. 138: 616, 2012.
Clot retraction: checks factor I, platelet count, and a particular platelet function (missing in Glanzmann's disease)
Urea solubility: checks factor XIII ("fibrinoligase", cross-links fibrin strands, rendering the clot insoluble)
Fibrin degradation products (FDP, "fibrin split products", FSP, X-Y-D-E): increased levels suggest ongoing fibrinolysis by plasmin, particularly in disseminated intravascular coagulation (DIC) or hyperfibrinolysis; these are also likely to be elevated when a large thrombus is present. This old test has been mostly replaced by D-dimer.
D-dimer: From broken-down clots that have been crosslinked by factor XIII. Once foolishly promoted as "a more specific test for DIC", it's now clear that D-dimer is elevated whenever there is intravascular thrombolysis, whether from DIC or big thrombi. (Confirmed once again: Am. J. Med. 123: 17, 2010). It's also up in liver failure as the liver can't clear it. * Future pathologists: Just how specific it is depends entirely on where you set your cutoff point.
* "Thrombin generation assay" is a high-tech test that measures the speed and intensity at which a clot forms from platelet-poor plasma in a special machine. It's supposed to pick up both bleeding and clotting tendencies not detectable by other assays (Am. J. Clin. Path. 139: 370, 2013.)
Platelet function testing: testing response to ADP, thrombin, collagen, serotonin, catecholamines, and thromboxane A2 -- all "platelet agonists". Platelet function testing explained: Arch. Path. Lab. Med. 126: 133, 2002. Remarkably accurate new assays: Am. J. Clin. Path. 122: 178, 2004.
Mixing studies: Mix the patient's blood half-and-half with blood from somebody who is healthy. If the patient has an antibody against a clotting factor, the tests will remain abnormal. If the patient is missing something important, the tests will become normal.
Tip: Get a functional, rather than an immune-based, assay for proteins C, S, AT-III, etc., etc. Some people's factors may contain a mutation that renders them non-functional but still demonstrable by immune methods.
Thromboelastrography: Blood is rotated in a cup and a sensor measures how fast it clots and how tough the clot is. Probably the best way to check for hypercoagulable blood in trauma patients -- i.e., who needs clotting factors, who clearly doesn't (J. Trauma 67: 266, 2009). Thromboelastometry goes to war (to guide thrombus prevention in wounded soldiers) -- J. Trauma. 69(S1): S40, 2010. Less helpful in the cardiac surgery unit: Am. J. Clin. Path. 142: 492, 2014.
INCREASED VASCULAR FRAGILITY ("nonthrombocytopenic purpuras", etc.): bleeding problems despite normal platelet count, bleeding time, PT, aPTT, TT, FDP)
Bleeding from fragile vessels, with normal platelets and coagulation, is seldom serious.
Massive bleeding is rare. Vascular fragility is more likely to cause skin bruises, dependent petechiae, gum bleeding (eating, toothbrushing), hematuria, nosebleeds, GI bleeds.
Causes include:
INFECTIONS (meningococcemia, gonoccemia, scarlet fever, SBE, other forms of sepsis (TNF damages the
endothelium; there must be other molecules as wel), rickettsial disease,
the many viral hemorrhagic fevers, Ebola![]() being only one) that particularly damage endothelium. The bleeding usually won't be massive,
but it warns of grave systemic illness.
being only one) that particularly damage endothelium. The bleeding usually won't be massive,
but it warns of grave systemic illness.
The reasons for bleeding in hemorrhagic fevers (including dengue, which is very common in much of the world) are still being worked out.
AMYLOIDOSIS
COLLAGEN PROBLEMS: scurvy, Ehlers-Danlos syndrome ("the human pretzel"), Cushing's syndrome, osteogenesis imperfecta (patients do not convert reticulin to regular collagen normally), hereditary hemorrhagic telangiectasis (Osler-Weber-Rendu disease; patients have an abnormal type III collagen or some related mutation and some vascular malformations that bleed easily -- this is the most serious bleeding problem in the group), "old age" ("senile purpura")
{05940} scurvy, gums
{38195} scurvy case, bone
IMMUNE-COMPLEX DEPOSITION IN VESSEL WALLS: serum sickness, Henoch-Schonlein purpura, cryoglobulinemia, wonder drugs (the usual cause of "leukocytoclastic vasculitis", lots of living and dead neutrophils around the cutaneous vessels, which in turn is the common cause of "palpable purpura" on the skin)
{12261} erythema multiforme case with purpura
{12262} erythema multiforme case with purpura
{12529} erythema
multiforme case with purpura
* MIND PHENOMENA (Gardner-Diamond autoerythrocyte sensitization / psychogenic purpura, religious stigmatization; do you believe in this stuff? I don't know....)
As noted, the usual platelet and coagulation tests are normal. (Capillary fragility tests using blood pressure cuffs, the old "tourniquet test", etc., are of limited sensitivity and specificity.)
REDUCED PLATELET NUMBER ("THROMBOCYTOPENIA"): Ped. Clin. N.A. 51: 1109, 2004, lots more
Platelets are the first line of defense against bleeding, plugging up little holes in capillaries (PRIMARY HEMOSTASIS) in seconds. Of course, they also help initiate coagulation (to solidify the blood before it gets through the bigger holes), and eventually become an important component of most clots. The activating of the clotting factors, which takes minutes, is called SECONDARY HEMOSTASIS.
Think of platelets as little band-aids that turn into bricks (fibrin's the mortar).
Excessive bleeding due to platelet disorders is apparent almost immediately after trauma. Excessive bleeding due to coagulation factor deficiency becomes apparent only after a few minutes.
The greatest concern in thrombocytopenia is intracranial bleeding.
Thrombopoietin ("megapoietin") cloned and characterized: Nature 369: 533, 1994 -- a relative latecomer among the known hormones.
Here's how your body keeps your platelet count where it belongs. Your liver (mostly) produces your thrombopoietin, and most of it gets sopped up by the circulating platelets. Any that's left-over stimulates the production of more platelets.
Your lab can assay thrombopoietin: Eur. J. Hem. 61:119, 1998.
Thrombopoietin is now being used to make people make extra platelets prior to chemotherapy; these are harvested and re-transfused (Lancet 359: 2145, 2002).
* And we now have eltrombopag, an non-peptide oral thrombopoietin-receptor agonist, that's finding use in chronic ITP (Lancet 373: 641, 2009; Lancet 377: 393, 2011) -- works for leukemia and myelodysplastic syndrome too of course (Blood 114: 3899, 2009.)
* Tomorrow's high-tech blood bank may contain equipment that generates platelets from cell cultures, without a donor: Blood 121: 3319, 2013.
* Thrombin does some carving on thrombopoietin and perhaps this is why more platelets are produced when you start clotting. Stay tuned.
Always order a CBC, which includes a platelet count, on anybody who seems seriously sick.
Thrombocytopenia is said to be present when platelet count is less than 100,000/mcL. Platelet problems are often heralded by petechiae on the skin and mucosal surfaces.
Bleeding after trauma (surgery, etc.) can be a problem when platelet count is below 40,000/mcL.
Spontaneous bleeding likely to occur only when the platelet count is below 20,000 or so. (Petechiae and purpura randomly over the skin, blood blisters in the mouth, GI, GU, CNS bleeding.)
{11526} hemorrhage in thrombocytopenia (* "Sweet's syndrome" case)
Severe spontaneous bleeding may be expected when count gets below 10,000/mcL.
Causes of thrombocytopenia are many.
{13805} giant platelets, myelofibrosis case
Tip: If you see only a few platelets and many of them are large ("megathrombocytes"), the patient is probably turning out platelets very rapidly (i.e., they have not fully separated from one another), i.e., they are being destroyed peripherally. Of course, to be sure, you may want to do a bone marrow exam. If platelets are being destroyed peripherally, you will see many, young megakaryocytes with hypo-segmented nuclei.
* Pitfall: When remission of a leukemia is being induced, the fragmentation of the white cells may result in fragments that are interpreted by the automated cell counters as platelets, disguising a serious thrombocytopenia (Arch. Path. Lab. Med. 123: 1111, 1999). One more reason to look at the peripheral blood smears yourself.
Patients with platelet abnormalities have increased bleeding time, normal PT, aPTT.
To distinguish a thrombocytopenia of decreased production (few or non-maturing megakaryocytes) or increased destruction (increased megakaryocytes), examine the bone marrow.
If platelets are rapidly being destroyed and the marrow is making them overtime, giant platelets are often abundant in the peripheral blood.
ISOIMMUNE / ALLOIMMUNE THROMBOCYTOPENIA
Neonatal alloimmue thrombocytopenia: Fetal-maternal incompatibility, analogous to hemolytic disease of the newborn (antigen usually HPA-1a which Dad had and Mom didn't; there are a few uncommon ones), except that it tends to happen during the first pregnancy by that father.
Mother's IgG antibodies against some specific platelet antigen on baby's platelets causes them to be destroyed. Very common (said to be ~1/600 US babies), usually mild (platelets stay above the dangerous range), but can be deadly (intracranial bleeds in utero). Read all about it: NEJM 337: 32, 1997.
* Mom is most likely to produce the antibody if she has HLA class III DRB3*0101.
Post-transfusion: After a PlA1-negative patient receives someone else's PlA1-positive platelets (to which the patient must be already sensitized), she starts destroying her own platelets. (Immune complexes adsorbed to the patient's platelets are probably the cause.)
IDIOPATHIC THROMBOCYTOPENIC PURPURA ("ITP", autoimmune thrombocytopenic purpura, "ATP", etc., review Blood 113: 6511, 2009). A group of diseases in which there are antibodies against platelets (assaying these antibodies is still problematic) and megakaryocytes (i.e., both increased destruction and decreased production).
ACUTE ITP: a disease of children, most often following a viral infection.
Platelets become coated with antibodies (sometimes anti-platelet autoantibodies, more often immune complexes from the viral illness) and get eaten by the RE system.
Thrombocytopenia in AIDS results from this mechanism, and/or autoantibodies against IIb-III and/or HIV attacking megakaryocytes that are positive for CD4.
CHRONIC ITP: a disease of adults, especially those with autoimmune disease. True anti-platelet antibodies are present (often against IIb-IIIa and/or Ib-IX), and platelets are destroyed by the RE system as in acute ITP. Some labs test for these antibodies but this isn't much used (Lab Med. 42: 687, 2011.)
* Romiplostim ("Nplate"), the molecule that stimulates the thrombopoietin receptor, seems to work wonders for chronic ITP: Lancet 371: 395, 2008; Blood 114: 3748, 2009. Eltrombopag: Lancet 373: 1562, 2009.
Both types have increased IgG in the platelet fraction. Splenectomy is necessary in some cases. (The spleen makes much of the offending antibody, and it eats most of the sensitized platelets.)
* Rituximab to suppress antibodies seems to help chronic ITP in adults: Blood 121: 1928 & 1976, 2013; less favorable Lancet 385: 1653, 2015. Adult immunization does not cause adult ITP: Blood 120: 493, 2012.
THROMBOTIC THROMBOCYTOPENIC PURPURA (TTP; review Am. J. Clin. Path. 121 S: S-89. 2004)
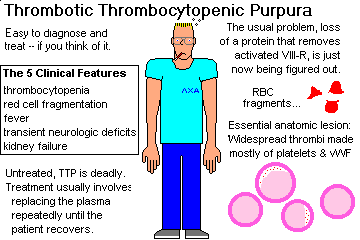
This is a dread disease that kills young adults. It is characterized by thrombocytopenia, microangiopathic hemolytic anemia (i.e., DIC), fever, transient neurologic defects, and renal failure.
Microthrombi occur in the arterioles and capillaries of most organs. They're usually worst in the heart. They are made of loose "hyaline" aggregates of platelets and von Willebrand's factor, with just a little bit of fibrin.
The pathophysiology yielded up its secrets with the discovery of von Willebrand factor cleaving protease (which breaks up big aggregates of von Willebrand's factor before they cause massive aggregation of platelets).
Familial TTP (* "Schulman-Upshaw") features a familial deficiency of this protease (NEJM 339:1578, 1998; gene ADAMTS-13 / vWF metalloprotease; Nature 413: 438, 2001; Blood 102: 1148, 2003; Blood 104: 2081, 2004; there's more to the story as the deficiency is prothrombotic but not sufficient to make the disease inevitable Blood 107: 3161, 2006, which make sense because if it were, the children would not be born). Alleles of varying severity Blood 120: 440, 2012.
TTP may be secondary to lupus (autoantibody of course... treating it South. Med. J. 101: 943, 2008), an unexplained antibody (depleting the antibody with bortezomib NEJM 368: 90, 2013), HIV-infection (South. Med. J. 88: 82, 1995), * ticlopidine (as after coronary stenting, JAMA 281: 806, 1999) and others.
Today you may diagnose it by the finding of severe thrombocytopenia plus microangiopathic hemolysis without alternative explanation; your pathologist can help by finding big aggregates ("ultralarge multimers") of vWF in the blood (Am. J. Clin. Path. 121-S: S89, 2004).
* Recombinant ADAMTS13 is now available Blood 119: 6128, 2012.
* "Thrombotic microangiopathy of unknown cause" is seen especially in patients given such treatments as mitomycin, cyclosporin, tacrolimus (South. Med. J. 101: 744, 2008), heavy-duty chemotherapy, quinine (with the antiplatelet syndrome), organ transplants, or total-body radiation. The microthrombi can appear either systemically (mimicking TTP) or mostly in the kidney (mimicking HUS), often long after exposure. The mechanisms aren't known, but probably involved damage endothelium. It would seem wise to not to call these either TTP or HUS. Overview of the syndromes of thrombotic microangiopathy: NEJM 371: 654, 2014.
If you are confused about how to tell TTP from HUS, especially in an adult, before autopsy, you are not alone -- in fact, NOBODY knows how to make the distinction, and today you'll hear the term TTP-HUS.
{08059} petechiae on heart, leukemia case
{21433} petechiae from football mouth-guard, no hemostasis problem
DEFECTIVE PLATELET FUNCTION (normal platelet count, prolonged bleeding time)
What platelets do for you:
Step 1: ADHERE (i.e., to collagen and basement membrane)
Step 2: RELEASE their ADP (makes platelets work better) and their catecholamine (local vasoconstriction) and their thrombin (activates fibrin directly)
Step 3: AGGREGATE (i.e., stick tight and recruit more platelets, due to ADP)
Step 4: FUSE tight to fibrin and each other, to seal the leak
Step 5: RETRACT (i.e., pull the clot tight)
Note that as long as platelet release and fibrin production are reasonably normal, there will not be bleeding from small nicks (i.e., shaving) even in patients with hemophilia.
* Lab workup of platelet disorders: Arch. Path. Lab. Med, 126: 133, 2002.
Hereditary:
DEFECTS OF PLATELET ADHESION
Bernard-Soulier disease (the most common of the several "giant platelets syndrome"; Arch. Path. Lab. Med. 131: 1843, 2007)
An autosomal recessive, thankfully-uncommon, severe bleeding disorder.
Giant, useless platelets that won't stick to the subendothelium; severe bleeding.
Bernard-Soulier patients lack glycoprotein Ib (GpIb) that binds the factor (VIII:R) that enables platelets to interact to collagen.
Stem cell transplantation for cure: Ann. Int. Med. 138: 79, 2003.
There is a mild dominant allele (big platelets, lowish counts, harmless): Blood 97: 1330, 2001; Am. J. Med. 112: 742, 2002. Some Bernard-Soulier heterozygotes have huge platelets but no problems (Blood 97: 1330, 2001).
There are at least 11 other genetic giant platelet disorders. All are even less common than Bernard-Soulier. The curious may read about the dominant forms in Am. J. Hem. 74: 254, 2003.
Von Willebrand's disease (see also below): Review Medicine 76: 1, 1997.
An autosomally inherited, qualitative or quantitative lack of Von Willebrand's factor.
There is no aggregation in response to ristocetin, but there is normal aggregation in response to epinephrine, collagen, ADP.
The platelet defect is corrected by normal or hemophilic ("factor VIII-deficient") plasma.
DEFECTS OF PLATELET SECRETION (of prostaglandins and ADP, the "release reaction"): various "aspirin-like" hereditary diseases too rare and complicated to outline here!
* Storage-pool disease. Some folks have absent granules. Gray platelet syndrome: No alpha granules. Hermansky-Pudlak: No delta granules. Chediak-Higashi: Diminished granules and some platelet dysfunction. Leave this arcane stuff to the hematologists.
* Hermansky-Pudlak syndrome: Any of several genetic diseases with defective production of melanosomes, absent delta platelet granules, and lysosomes (Blood 96: 4227, 2000; Blood 107: 4857, 2006; Am. J. Path. 166: 231, 2005); patients are albinos and in many variants, pulmonary fibrosis causes death.
* Gray-platelet disease: No granules. Autosomal-variable, and a bleeding problem. Thankfully rare. See Blood 98: 1382, 2001.
Of course, today we induce platelet dysfunction using meds such as aspirin and the ADP-receptor binders for our coronary artery patients (update NEJM 361: 940, 2009).
DEFECTS OF PLATELET AGGREGATION
Thrombasthenia (Glanzmann's disease, formerly "tired platelet syndrome"):
An uncommon, autosomal recessive, severe bleeding disease.
Platelets fail to aggregate on an un-anticoagulated peripheral smear, with ADP, collagen, epinephrine, or thrombin. Clot retraction is also absent. (They lack the integrin glycoprotein GPIIb-IIIa that binds fibrinogen which in turn bridges platelets. Pathologists stain this with CD41 and CD61 -- megakaryocytes failing to stain well indicates Glanzmann's. Read all about it: Am. J. Hum. Genet. 53: 140, 1993; molecular biology Blood 90: 669, 1997.) An acquired autoimmune variant exists (J. Ped. 144: 672, 2004).
* Gene therapy for Glanzmann's works in mice: Blood 106: 2671, 2005.
Acquired defects:
Aspirin permanently inhibits cyclooxygenase (by acetylating it), preventing production of thromboxane A2 and producing an acquired secretion defect that lasts for the life of the platelet (a platelet lives about 9 days).
The other NSAIDS temporarily block cyclo-oxygenase.
* All about NSAIDS and platelets: Am. J. Med. 106 (5B): 25S, 1999.
In uremia, there is a complex platelet defect reversed by dialysis, which may be due to * phenol and/or * guanidinosuccinic acid.
* Alcohol enhances the effect of aspirin on platelets. During an alcoholic binge, the platelet numbers drop; during alcohol withdrawal, there is a rebound thrombocytosis that can produce a stroke if somebody is predisposed.
|
|
THROMBOCYTOSIS
THROMBOCYTOSIS is a platelet count above 400,000/mcL.
It may occur as a rebound after severe bleeding, after surgery or sepsis, following splenectomy, or just runs in the family (JAK2 kinase domain mutations Blood 123: 1372, 2014), or is part of the disease in iron deficiency (common, but no one knows why), primary liver cancers that make thrombopoietin (Dig. Dis. Sci. 58: 1790, 2013). carcinomatosis (most famously ovarian cancer, interleukin 6 effect NEJM 366: 610, 2012), or * Hodgkin's disease; update on the molecular mechanisms of paraneoplastic thrombocytosis Blood 124: 184, 2014.
THROMBOCYTHEMIA is a sustained platelet elevation over 800,000/mcL. Unless there's some other obvious explanation, this indicates some myeloproliferative disorder (leukemia, polycythemia vera, early myelofibrosis, etc).
If there is no other known illness, look for a JAK2 mutation -- this now helps define "essential thrombocythemia", a myeloproliferative disorder that nowadays does not shorten life expectancy. Patients may have no problems, or they may have thrombi, or their platelets may not work (an extremely high count depletes vWF.) Or platelets plugging vessels may cause brain ischemia, or erythromelalgia (a skin pain syndrome). Even then, it's likely to remain asymptomatic for a long time, and is seldom lethal (Am. J. Med. 117: 755, 2004; Arch. Path. Lab. Med. 130: 1144, 2006). Update Mayo Clin. Proc. 80: 97, 2005.
You're familiar with JAK2 which bears a signature mutation in the large majority of cases of polycythemia vera, primary myelofibross, and essentiual thrombocythemia. For each, there may instead by a mutation in calreticulin (CALR; Blood 124: 3964, 2014).
* Future pathologists only: Look closely at the marrow megakaryocytes. If you see them loosely clustered, with "staghorn" nuclei, and/or hyperlobulated nuclei, think especially of essential thrombocythemia (Am. J. Clin. Path. 136: 618, 2011).
You'll learn how to manage these folks. Often the best treatment is nothing. The great wonder drug of the 1990's was anagrelide. Hydroxyurea plus low-dose aspirin may work even better: NEJM 2005: 353, 2005. Today, the JAK2 inhibitors are taking over.
* A variety of preleukemia syndromes feature excess platelets, most notably the "refractory anemia with ringed sideroblasts with marked thrombocytosis" group (Am. J. Clin. Path. 135: 398, 2011).
{24786} essential thrombocythemia
* {13748} "megakaryocytic myelosis"
ABNORMALITIES IN COAGULATION FACTORS (abnormal PT, aPTT, TT, and/or FDP; usually normal platelet count and bleeding time)
Deficiencies of all of the clotting factors have been described.
For each, the deficiency can be mild or severe, the protein can be absent or just defective, or the deficiency may be due to an inhibitor (antibody, etc.) against it.
Deficiencies may be hereditary or acquired:
Hereditary deficiencies involve a deficiency of a single factor.
Acquired deficiencies (except those due to an autoantibody) involve several factors
Deficiency of vitamin-K-dependent factors (neonates, malabsorption, heavy antibiotics, coumarin therapy, bad liver trouble).
Remember the body requires vitamin K so that the body can synthesize can make gamma-carboxyglutamic acid, present in factors II, VII, IX, and X, protein C and protein S.
Of these, factor VII is usually the first to go, so the defect will appear initially in the extrinsic pathway, i.e., abnormal PT.
Heparin therapy potentiates antithrombin III, so heparin indirectly inactivates thrombin.
* The thrombin time estimate for fibrinogen is useless in the heparinized patient, so the lab uses snake venom ("Russell viper venom time, RVVT") instead of thrombin.
* An ultra-rare cause of bleeding is systemic mastocytosis -- when the mast cells degranulate, the person becomes hypotensive from the histamine, and gets a big dose of endogenous heparin as well.
"Circulating anticoagulant" is the common term for any abnormal protein, not part of the normal clotting-anti-clotting systems, that interferes with coagulation.
Such a protein may be either an autoantibody against a clotting factor, or an inhibitor of one or more steps. Both are common enough in systemic lupus, rare in other people.
Disseminated intravascular coagulation (all factors consumed, as are platelets.)
HYPERFIBRINOLYSIS
Surgeons describe "acute coagulopathy of trauma" in the severely injured; at least part of the problem is activation of plasmin (for some reason) during the first hour (Ann. Surg. 252: 434, 2010). It is treated with recombinant activated factor VII, and now considered preventable by using more fresh-frozen plasma during resuscitation (Am. J. Clin. Path. 137: 566, 2012).
Of course this is seen when thrombolytic plasminogen activators are given for heart attack or stroke; this is one of the most feared consequences.
* Congenital causes of hyperfibrinolysis, such as absence of alpha-2-antiplasmin or plasminogen activator inhibitor type I, are thankfully rare. When the liver fails, hyperfibrinolysis is just one more problem.
Patients with coagulation factor deficiencies rarely have spontaneous petechiae or purpura. Instead, they get ecchymoses or hematomas after minor injury that the platelets don't handle. Bleeding for days after tooth extractions, or bleeding into joint spaces (hemarthroses) are common.
DEFICIENCIES OF FACTOR VIII COMPLEX
Factor VIII:C (procoagulant) is the clotting factor required to activate factor X in the intrinsic pathway. It is coded on the X-chromosome.
It circulates bound to VIII:R (von Willebrand's factor, made in endothelium and megakaryocytes) which is required for the interaction of platelets with subendothelial collagen and also protects VIII:C from destruction.
VIII:R is required for platelet aggregation by ristocetin.
Von W's factor is a tumor marker for Kaposi's sarcoma (of endothelial origin) and other angiosarcomas.
|
CLASSIC HEMOPHILIA ("factor VIII deficiency", hemophilia A, "royal blood"): Review of the two major hemophilias:
Lancet 379: 1447, 2007; BMJ 344: e2707, 2012.
Sex-linked recessive deficiency of factor VIII:C (gene F8). This is a mild, moderate, or severe bleeding disorder affecting 1 in every 5000 men. Female carriers may have mild disease due to unlucky lyonization (Blood 85: 599, 1995). Severe cases have maybe 1% or less activity of the healthy protein. |  Queen Victoria actually carried IX deficiency |
* Total knee replacement has become commonplace in patients with hemophilia (J. Bone Joint Surg. 92: 1085, 2010).
At least in the developed nations, most of these men now lead near-normal lives thanks to recombinant factor VIII. Contact sports participation (American football, wrestling) is okay even for severely affected boys (JAMA 308: 1452 & 1480, 2012).
* Managing these patients is expensive but works; keys are activated factor VII (Blood 102: 2358, 2003; update Blood 120: 891, 2012) and inducing tolerance (Blood 96: 1698, 2000).
Gene therapy for hemophilia should be with us in the next decade or so. It has already worked in animals (Blood 102: 2031, 2003; Blood 106: 1552, 2005; stem cells cure pigs Blood 107: 3859, 2006). More new approaches: Blood 114: 526 & 667, 2009; J. Clin. Inv. 119: 2086, 2009; Blood 114: 526, 2009. Transferring to humans hasn't been so successful as we'd hoped, but something's bound to work. An earlier attempt using fibroblasts, without even a virus, raised levels; the trick is to get the fibroblasts to continue making the factor VIII for more than a few months (NEJM 314: 1735, 2001). Preventing antibodies against factor VIII in a mouse model: Blood 108: 19, 2006.
VON WILLEBRAND'S DISEASE ("pseudohemophilia")
By far the commonest inherited hemorrhagic disorder. About 1% of humankind is affected. It involves a qualitative or quantitative deficiency of VIII:R/vWF (thus often also low VIII:C as it's not protected) and/or the platelet factor to which it binds (* glycoprotein Ib α).
Thus there is prolonged bleeding time and, in severe cases, some prolongation of aPTT. And of course the platelets don't stuck together well; in particular, they fail to respond to ristocetin, which should active the vWF receptors on platelets) resulting in the vWF gluing the platelets together. If you don't understand this, please review it.
Autosomal inheritance varies according to subtype: dominant (asymptomatic to moderate forms) or recessive (severe forms; carriers are asymptomatic).
All the von Willebrands' syndromes are autosomal dominant and relatively mild, with the exception of type III, which results from two doses.
Easy...
II. Mutant vWF
IIb. Large complexes bind inappropriately to platelets and megakaryocytes (Blood 108: 2587, 2006), which are then cleared; thrombocytopenia; increased reactivity to ristocetin; don't use desmopressin (why not?)
IIn. Binds to platelets much better than to VIII; low VIII levels
III. vWF is normal, but very little is made. Autosomal recessive, severe illness
To screen, ask for a von Willebrand's profile -- a vWF antigen, a vWF activity level, and a factor VIII activity level.
In a woman with heavy periods and a normal pelvic exam, von Willebrand's is quite likely: Lancet 351: 485, 1998.
Sorting out "mid von Willebrand's with excessive bleeding after a minor accident" from inflicted trauma is an extra challenge in an already-contentious area: Forensic Sci Med. Path. 7: 37, 2011.
Remember that von Willebrand's factor is an acute phase reactant, so levels may be normal during other illnesses.
Sex hormones (especially estrogens) partially correct the molecular deficiency in some types, so the disease often gets better at puberty; don't trust a screening test during pregnancy. The management of von Willebrand's disease, especially before surgery, used to be based on administering factor VIII concentrates, which are full of good vWF multimers. Treatment was revolutionized in the 1980's by the discovery that desmopressin (!) raises vWF levels.
ACQUIRED VON-WILLEBRAND'S has at least three etiologies.
Think also of autoantibodies, and adsorption to the surfaces of tumor cells (infamously Wilms' tumors and the Ewing sarcoma family; Mayo Clin. Proc. 77: 181, 2002).
Click here for Uncle Sam's guidelines (2008) on diagnosing and managing this most common of bleeding disorders.
FACTOR IX DEFICIENCY (hemophilia B, Christmas disease)
Sex-linked recessive deficiency (gene F9), often even more severe than, but otherwise similar to, classic hemophilia. One man in 25,000 is affected. Again, the joint disease is the most troublesome feature from day to day.
Mr. Christmas was the first patient discovered to have factor IX deficiency.
Like patients with factor VIII deficiency, most of these people now enjoy near-normal lives, the recombinant protein having been introduced in 1999. Only about 3% make troublesome antibodies against factor IX.
* Attempts at cure with gene therapy are of course ongoing. First reports of major success: NEJM 365: 2357, 2011; Blood 119> 2973 & 3038, 2012.
This is the illness passed by Queen Victoria to some of her family. It enabled the identification of the bones of Prince Alexei Romanov (Proc. Nat. Acad. Sci. 106: 5258, 2009)
* Factor VII "deficiency" discovered in African-Americans is often a non-disease, with no bleeding but a long PT. Their factor VII does not respond to the rabbit reagent used in the test, but works normally (Am. J. Clin. Path. 126: 128, 2006).
* Alpha-2 plasmin inhibitor (antiplasmin) deficiency has been described; you remember this is the stuff that binds up any active plasmin that makes it into the flowing blood (Br. J. Hem. 114: 4, 2001). The treatment is to administer tranexamic acid or epsilon-amino caproic acid, both of which prevent binding of plasminogen to fibrin.
Lots more.
* IATROGENIC BLEEDING DISORDERS
Most recently, apixaban (the orally-active inhibitor of Xa that helps prevent venous thromboemboli in orthopedic surgey patients, etc.) was tested along with aspirin in patients after acute coronary events. The study had to be ended early because there was no benefit and patients in the drug group hemorrhaged (NEJM uly 24, 2011).
HYPERCOAGULABLE BLOOD: "Thrombophilia"; "hypercoagulopathy". Not rare, but tends to get overlooked. Big reviews: Am. J. Med. 116: 81, 2004; Ann. Int. Med. 138: 128, 2003; Postgrad. Med. 101(5): 249, May 1997. Lab screening: update Am. J. Clin. Path. 126: 120, 2006, Am. J. Clin. Path. 137: 553, 2012, and J. Clin. Path. 59: 156, 2006 (nobody really knows what to order or when to order it).
* Lupus anticoagulant, factor V Leiden, prothrombin G20210A, and protein S deficiency in particular seem to place a woman at increased risk for fetal loss: Lancet 361: 901, 2003.
* Thal intermedia -- seems to have just the wrong mix of activated platelets and hemolyzed red cell stroma
* It's finally here... a "genome-wide screen" comparing the genes from people with DVT's and those without. Dozens of point mutations lurk within genes, familiar and unfamiliar: JAMA 299: 1306, 2008.
Acquired:
* Prothrombin G20210A is a point mutation affecting 2% of people; it renders blood slightly more coagulable. It is insufficient, by itself, to cause thrombosis. It can be detected only by DNA testing and only recently have people started talking about this being worthwhile (Arch. Path. Lab. Med. 126: 1319, 2002, contrast Mayo Clin. Proc. 75: 595, 2000; whether it's worth testing for, it's real: Am. J. Clin. Path. 127: 68, 2007).
PROTEIN C DEFICIENCY (Blood 85: 2756, 1995) and PROTEIN S DEFICIENCY are relatively common; 1 person in 300 is heterozygous for lack of protein C. (The most severely affected homozygotes get lethal purpura fulminans as babies.) You know that thrombomodulin on intact endothelium activates protein C when it meets thrombin (why is that good?), and that S and C work together to destroy Va and VIIIa. These patients (notably the homozygotes, protein C deficient heterozygotes are at around 8x increased risk, homozygotes 80x: Lancet 341: 134, 1993) clot their blood too readily, and are prone to pulmonary emboli and so forth. Both protein S and protein C are vitamin K dependent proteins. Thus the benefits of warfarin therapy are probably limited; this is recently supported (Arch. Int. Med. 157: 2227, 1997); since warfarin depresses protein C levels before it depresses II, VII, IX, and X, these people (or even apparently normal people whose protein C is inhibited before II, VII, IX and X) may actually develop skin necrosis ("warfarin necrosis") from taking the medication; see Ob. Gyn. 90: 671, 1997; Br. J. Surg. 87: 266, 2000.
HEREDITARY DEFICIENCY OF ANTITHROMBIN III is quite common. When it's just a matter of too little being produced (* type I AT3 deficiency), there's an increased risk especially for deep vein thrombi. When it's mutated (* type II AT3 deficiency), perhaps the patient will not respond to heparin (which works by enhancing the effect of normal AT3).
We've already probed the mysteries of ANTIPHOSPHOLIPID ANTIBODY SYNDROME (Blood 86: 617, 1995; Am. J. Med. 100: 530, 1996; mega-review Lancet 353: 1348, 1999; Arch. Path. Lab. Med. 126: 1326, 2002).
Two different types of antiphospholipid antibodies are described:
* Despite the popular name "lupus anticoagulant", if the patient does not already have lupus, discovering the blood factor does not predict development of the illness (Medicine 84: 225, 2005).
Should you anticoagulate them all (NEJM 332: 993, 1995)? Give them low-dose aspirin? Do nothing unless they're very sick (Am. J. Ob. Gyn. 176: 1099, 1997)? Nobody knows yet what's best.
It seems that the antibodies activate endothelial cells (Circulation 99: 1997, 1999).
We're discovering that many of them also make autoantibodies against proteins C and/or S, and the autoantibody itself may induce TF on the surfaces of monocytes, etc. (Lancet 350: 1491, 1997).
![]() Antiphospholipid antibody
Antiphospholipid antibody
Lost baby
Pittsburgh Pathology Cases
ACTIVATED PROTEIN C RESISTANCE is usually caused by a particular point mutation of factor V (V LEIDEN). It is a common, hated cause of thrombosis, pulmonary emboli (NEJM 336: 399, 1997; about half of young folks with "unexplained" DVT's have it), second-trimester miscarriage / preterm birth (Lancet 358: 1238, 2001), and accelerated atherosclerosis (NEJM 332: 912, 1995).
The activated form of this factor V resists the anticoagulant effect of protein C (J. Lab. Clin. Med. 125: 566, 1995). This was recognized in 1995 as the most common of the then-five known major hypercoagulability syndromes, affecting maybe 3% of the public. It also does not clear VIIa so well as normal Va does.
A major 1998 study found no benefit from long-term anticoagulation of V-Leiden people (BMJ 316: 95, 1998.)
Not surprisingly, V-Leiden is a significant coronary risk factor: Am. Heart J. 147: 897, 2004.
Stay tuned: HYPERHOMOCYSTEINEMIA resulting from any of several kinks in methionine metabolism, or perhaps even just lack of folate in the diet (stay tuned), is now known to be a serious risk factor both for accelerated atherosclerosis and venous thrombi. Update Arch. Path. Lab. Med. 126: 1367, 2002.
Homocysteine damages the endothelium, and does various things to various coagulation factors that are presently being worked out.
Mild forms may be extremely common (Lancet 345: 902, 1995; NEJM 334: 759, 1996; Am. J. Clin. Path. 108: 115, 1997). fortunately, you can treat it with vitamin B12 and folic acid.
Generally, the thrombophilias are serious risks for clots in the spiral and intervillous arteries of the pregnant uterus. This in turn places the pregnancy at risk for severe pre-eclampsia, abruption, fetal growth retardation, and stillbirth (NEJM 340: 9, 1999.)
Uh... Please don't work up thrombophilia immediately after the acute episode. The labs will be off (why?)
* The treatment of thrombophilias in general, and the prevention of post-operative thrombosis in particular, is likely to be revolutionized by various oral inhibitors of factor Xa -- most famously rivaroxaban. See Lancet 372: 31, 2008. By now there are several similar medications in the pipeline.
* Or maybe plain old aspirin is best after all for preventing recurrent deep vein thromboembolism -- NEJM 366: 1959, 2012.
DISSEMINATED INTRAVASCULAR COAGULATION ("DIC", defibrination syndrome; "Death Is Coming")
We have already reviewed this in "general pathology". This is an acquired deficiency of clotting factors and platelets; they are being used up.
RBC's also get shredded on the intravascular strands, producing helmet cells, schistocytes, blister cells, keratocytes, etc.
![]() DIC
DIC
Schistocytes, no platelets
Wikimedia Commons
DIC may be caused by:
RELEASE OF TISSUE CLOTTING FACTORS INTO THE BLOODSTREAM
obstetrical catastrophe (tissue factors from placenta or amniotic fluid embolism)
major tissue injury (burns, heat stroke, surgery, trauma)
acute promyelocytic leukemia (tissue factor from "pro"'s granules, plus a surface factor that activates plasminogen NEJM 340: 994, 1999)
mucinous adenocarcinomas (mucin activates factor X directly)
sepsis (tissue factor from PMN's)
* filovirus infection (Ebola![]() ,
Marburg) -- monocytes express tissue factor on their surfaces Lancet Inf. Dis. 4: 487, 2004.
,
Marburg) -- monocytes express tissue factor on their surfaces Lancet Inf. Dis. 4: 487, 2004.
snakebite
ENDOTHELIAL DAMAGE
Viral hemorrhagic fevers (famously Ebola)
vasculitis (especially malignant hypertension, meningococcemia, rickettsial disease)
Kasabach-Meritt syndrome (giant hemangioma with ongoing "localized DIC" inside)
hypothermia (as in cardiac surgery: poorly understood. Sem. Thorac. Card. Surg. 9: 246, 1997.)
 Kasabach-Merritt syndrome
Kasabach-Merritt syndrome
Pittsburgh Pathology Cases
"Chronic DIC" ("compensated DIC") may result from underlying malignancies (prostate cancer especially), myelodysplastic syndromes, or "idiopathic". The body may overcompensate by increasing the platelet numbers above normal, but the clotting factors remain relatively depleted.
The essential treatment of DIC is that of the underlying disease. White-knuckle hematologists have given anticoagulants to bleeding patients. The key is of course treating the cause. The management of disseminated intravascular coagulation has been revolutionized by the introduction of activated protein C (Br. Med. J. 327: 974, 2003).
{03178} DIC, kidney, gross with hemorrhages
{03180} DIC, glomerulus, with fibrin-platelet thrombi
{03189}
DIC, petechiae on heart
{03192} DIC, liver with recent infarcts
{09629} DIC, fibrin-platelet thrombus in brain
{39649} DIC,
fibrin-platelet thrombi
{39817} schistocytes
{13895} schistocytes
{12237} Kasabach-Merritt syndrome patient
Remember:
Defects of the extrinsic pathway (normal aPTT, prolonged PT) usually indicate early liver disease or coumarin therapy (congenital factor VII deficiency is rare)
 Factor VII deficiency
Factor VII deficiency
Pittsburgh Pathology Cases
Defects of the intrinsic pathway (normal PT, prolonged aPTT) include factor VIII and IX deficiencies or circulating anticoagulants (congenital factor XI and XII deficiencies are rare)
Defects of both pathways (prolonged PT and aPTT): usually indicate heparin or coumarin therapy, DIC, advanced liver disease, or circulating anticoagulants (congenital factor II, V, and X deficiencies are rare)
Defects of neither pathway (normal PT and aPTT): fragile vessels, platelet problem, or factor XIII deficiency (remember urea solubility test)
Cryoprecipitate contains fibrinogen, vWF, factor VIII, factor XIII, and fibronectin, but no factor IX.
The coagulation system, with factors, platelets, and vessels, is a chaotic one -- especially, it's probably impossible to predict the effects of large numbers of medications on ICU patients, who famously develop bleeding disorders and/or hypercoagulability (Crit. Care Med. 38(S6): S198, 2010).
 |
POSTSCRIPT: "INTELLIGENT DESIGN" AND "IRREDUCIBLE COMPLEXITY"
A few of the early Christian writers argued that the earth could
not be round. They used several fallacies, most famously
that people on the other side would fall off and that there would be nothing
to hold it up.
These writers claimed that all non-Christian thought was
deeply flawed and led to immorality and the world's evils.
The great church leader Augustine, who is probably best-known today
for his "Confessions", urged these people to stop. Every informed
person knew they were wrong -- ludicrously so -- and they were discrediting the Christian faith.
During the middle ages, Anselm, one of the archbishops of Canterbury,
came up with the "ontological argument" to prove the existence of God.
By definition nothing can be better than God, existing is better than not
existing, so God must exist. I doubt that many people have ever found this very
persuasive. Some of Anselm's own contemporaries didn't,
and Anselm replied graciously to those who disagreed.
From what I have read and heard,
the clotting cascade is the centerpiece of today's most-often-cited proof
of the existence of God. The claim that every portion of the clotting cascade
is finely-tuned to work together was popularized by Michael Behe,
the only "intelligent design" advocate at the national level with bona fide scientific
credentials. (I refuse to recognize the one other guy, whose scientific
training was sponsored -- with the intent that he would write creationist
books -- by a cult that has taught that its founder conceived his eight children
by blowing in his wife's ear. The other three major players are all
law-school types, who are professionally trained to argue positions
even when they know they are wrong. The mathematician also
refused at the last minute to testify.) Unlike
Archbishop Anselm, the intelligent design
proponents (though superficially polite)
accuse their opponents of the vilest motives
and of rank stupidity.
Real scientists will recognize the old creationist
fallacy, "How could A evolve without
B, and how could B evolve without A?" Of course, things evolve together.
And Behe's claim that the coagulation cascade is "irreducibly complex"
is indisputably false.
The evolution of the clotting cascade is well-documented,
and as Darwin's theory predicts, it seems to give the same
phylogenetic tree as classic comparative anatomy. (If this really failed
for even a
single protein or gene, Darwin's theory would be refuted and "intelligent design"
pretty much established.
Are people like Behe looking?
Of course not. The exceptions or can't-call cases, discovered of course
by real scientists, that you'll see cited in "the ongoing debate"
are obviously minor aberrations.)
Whales lack factor XII, the gene being inactivated.
Turtles lack factors XI and XII.
Fish lack prekallikrein, XI, and XII.
Lampreys have a primitive system with tissue factor, prothrombin,
and fibrinogen.
Obviously the rest of the cascade
developed unit by unit to modulate the primitive system.
And this totally refutes the idea of "irreducible complexity."
See PNAS 100: 7257, 2003.
Origins of the vertebrate coagulation system: Thromb. Hemo. 89: 420, 2003
(England); Blood Cell. Mol. Dis. 29: 57, 2002.
There's a review of all the vertebrate systems
in J. Thromb. Hemo. 1: 1487, 2003.
Conservation back to the horseshoe crab: J. Mol. Bio. 282:
459, 1998.
The common origin of the clotting and complement cascades
(like Darwin's finches, everything used to do something else):
Trend Bioch. Sci. 27: 67, 2002.
A theologically-inclined guy at Harvard reviews
this in J. Thromb. Hemo. 1: 227, 2003.
It is inconceivable that the "intelligent design" proponents
do not know this
by now, and the fact that they persist tells me a great deal about
who they really are. I wrote the above in 2003.
By November 2005, the principal "intelligent design"
website (the Discovery Institute, a front group for the
Unification Church / Moonies) mentioned the business about clotting in
lampreys, and provides a link
to a page by Behe on which the data is supposedly reviewed and the
argument is supposedly refuted. But the linked page doesn't even mention it.
You can find it yourself. This is typical of how disinformation artists operate.
In "Kitzmiller vs. Dover" (2005), Behe admitted under oath in open court
on cross-examination
that he
knew the blood clotting cascade argument to be false. As far as I am concerned (and the judge was
concerned), this ended the "intelligent design" business. The handful of other
scientists who had been named as witnesses all refused to testify.
The lawyering stopped, with the losers going on
to making political capital and money for ultraconservative churches
by claiming to be persecuted saints.
For people of science, and informed members of the public,
the whole business will be
remembered for what it really was -- one more disinformation
campaign by pseudo-religionists. Was the motive the promotion of public
virtue (i.e., the belief that people are better-behaved if they believe
these sorts of lies?) Or was it just a money-maker? As a close observer,
I believe the truth stands
somewhere in the middle.
One objection that I have to this stuff involves disease itself,
my focus. If God builds genes and living systems to order,
why not make a more stable genetic system, one that would not be
so subject to mutations? Every human being who suffers from hereditary
disease or from cancer is a reminder of the real origin of our bodies.
To his credit, Dr. Behe acknowledged in the fine print of his book, "The Edge of Evolution",
that all living creatures have a common descent ("common ancestry of chimps and
humans... Despite some remaining puzzles, there's no reason to doubt
that Darwin had this point right, that all creatures on earth
are biological relatives." Remember he's the only creationist to go under oath lately.
If you are involved with this "intelligent design / creationism" stuff, please stop.
If you (like me) are a person of faith, you should demand that people
stop spreading lies as "proof of the existence of God."
I find nothing "spiritual" or "moral"
about wholesale breaking of the ninth commandment.
The Christian Bible compares our present "animal bodies" to
our future state as spiritual beings.
Like Job, I prefer to stand in awe and not demand answers to everything
right now. I expect you do, too.
| New visitors to www.pathguy.com reset Jan. 30, 2005: |
 Drop by
and meet Ed Drop by
and meet Ed
|

|

| If you have a Second Life account, please visit my teammates and me at the Medical Examiner's office. |
Teaching Pathology
 Ed's Pathology Review for USMLE I
Ed's Pathology Review for USMLE I
 | Pathological Chess |
 |
Taser Video 83.4 MB 7:26 min |
 |
Click here to
see the author prove you can have fun skydiving without being world-class. Click here to see the author's friend, Dr. Ken Savage, do it right. |
